My Peripheral Brain
*
My Peripheral Brain *
"I consider that a man's brain originally is like a little empty attic, and you have to stock it with such furniture as you choose. A fool takes in all the lumber of every sort that he comes across, so that the knowledge which might be useful to him gets crowded out, or at best is jumbled up with a lot of other things, so that he has a difficulty in laying his hands upon it. Now the skillful workman is very careful indeed as to what he takes into his brain-attic. He will have nothing but the tools which may help him in doing his work, but of these he has a large assortment, and all in the most perfect order. It is a mistake to think that that little room has elastic walls and can distend to any extent. Depend upon it there comes a time when for every addition of knowledge you forget something that you knew before. It is of the highest importance, therefore, not to have useless facts elbowing out the useful ones."
~Sherlock Holmes

CC: CP - the Approach
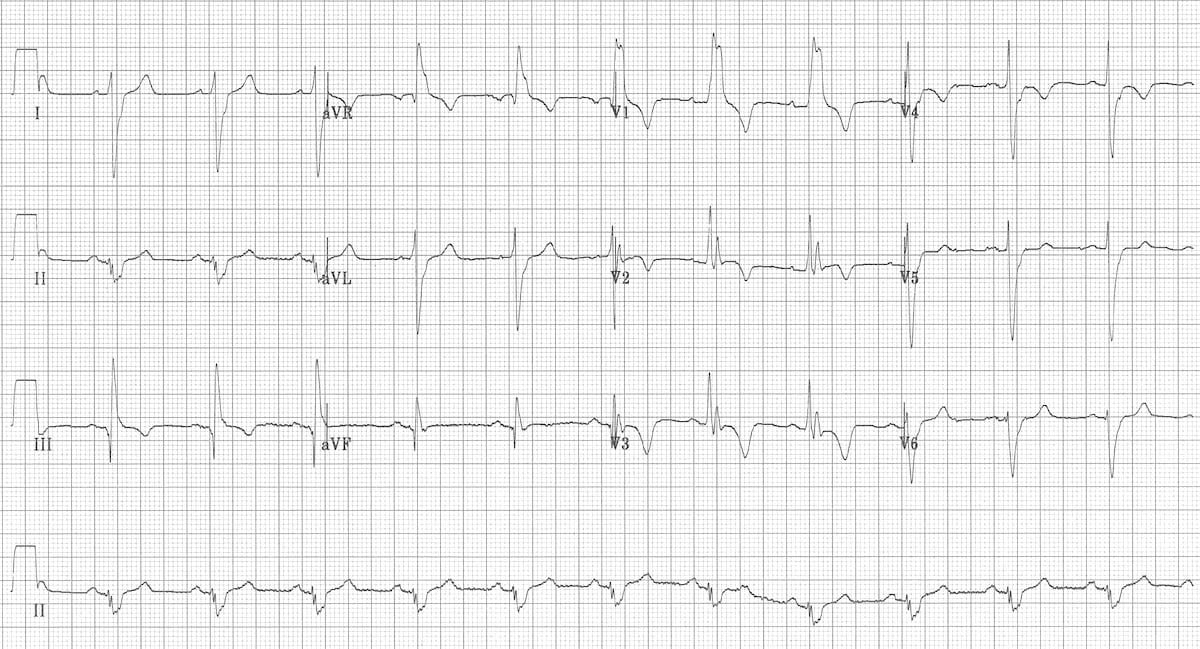
Is this a Levine sign???
If you have worked in the ER for one day, you have seen a patient with vague chest pain (CP). Unfortunately, they do not usually come in with a sign telling you what to do. Many times, the patient looks great, and you can predict that EKG and labs will be normal, but then there are times when I have been surprised when someone rules in for an NSTEMI or the EKG for the patient insisting it is just "heartburn" shows a STEMI.
And don’t forget the other dangerous causes of CP. I have seen a patient with a negative D-dimer almost get discharged home with a massive pneumothorax, and unfortunately, a patient code during a stress test due to an undiagnosed pericardial effusion with pericarditis, not CAD, the cause of his symptoms. Recently, with extremely long wait times and work-ups initiated from triage, I have found several pneumonias and PEs in patients waiting hours for serial troponins to “rule out” ACS.
For this reason, I argue for a scripted approach to all chest pain, and what better way to do that than with a CP template???
Your ED likely has protocols (i.e. scripts) to follow for acute MI getting to the cath lab in under 90 min and order sets for NSTEMI. Let's create a script we can follow to ensure we never miss a life threat, consider even rare diagnoses, and most importantly document a thorough chart that minimizes the risk of ever being successfully sued in a rare, atypical, or subtle case of CP.
To accomplish this, I suggest an order or a method to my madness that does not follow the typical order taught in school. Sure, we will get to the history of present illness (HPI) and review of systems (ROS), both of which we no longer need to document in excruciating detail for billing purposes, yet are still important. Instead, I propose a more realistic order as seen below. In every ER where I have worked, patients with CP get an EKG very early on in the visits, so I suggest that you look at this first.
Overview:
First, look at the ECG
Is it a STEMI?
An NSTEMI?
Other important ECG patterns to recognize
Next, what are possible Immediate life threats: Aortic dissection, Pulmonary embolism, Tension pneumothorax, Tamponade
What else to include in your Differential? Pericarditis and other non-emergent stuff.
Next, check the vital signs - What clues can you pick up (or miss) on VS?
Interviewing the patient? Yeah, the history still matters — What questions to ask.
Examining the patient - What are the key components of the exam to include on every CP pt?
Does the PE even matter?
BONUS: How to incorporate bedside ultrasound into every CP evaluation
Decision time: Medical Decision Making and tools.
Labs, tests, and other things to order (or not order).
HEART pathway for low-risk chest pain
Well’s, PERC, and others
Putting it all together: The full CP template
Bonus: How to not get (successfully) sued on a CP case!!!
Before we jump to my typical workflow, let’s review the “Don’t miss Diagnoses”.
CC: CP - the ECG (STEMI)
ECG
The ElectroCardioGram
Obviously, almost everything in EM cardiology starts with an EKG, and we also know that anyone with a chest and pain somewhere in the body will get an EKG from the triage nurse. Just think of that last patient you saw with finger pain who said, "I read online that this could mean a heart attack.”
In my experience, the difficulty in cardiac patients in the ED is not in those that present with a grossly abnormal EKG showing a STEMI. These patients can be diagnosed by EMS and essentially, present with a disposition. Give aspirin and your hospital's preferred antiplatelet, call cardiology, and ship them to the cath lab. No, the challenge in emergency medicine is patients without obvious ischemic findings on their EKG, figuring out who to work up and pursue further testing, and keeping in your differential all the other important cardiac pathologies that could masquerade as other diseases.
Not that you are allowed to miss a STEMI, so here is a quick review below, along with other STEMI equivalents or subtle changes that can indicate myocardial injury.
The ST-segment Elevated Myocardial Infarction (STEMI)
Figure 8. Electrocardiogram example of ST-segment elevation. The initial onset of the Q wave shown by arrow 1 serves as the reference point and arrow 2 shows the onset of the ST-segment or J-point. The difference between the two identifies the magnitude of displacement. Measurements of both arrows should be made from the top of the electrocardiogram line tracing.
STEMI is described in the Fourth Universal Definition of Myocardial Infarction as the following (in the absence of LVH or BBB)
New ST Elevation at the J point of at least 1 mm in two contiguous leads (except for V2-V3, see below)
In leads V2-V3, ST elevation at the J point must be
≥2 mm in men over 40 or ≥2.5mm in men under 40 y/o
≥1.5mm in women regardless of age
Wait, what are contiguous leads??? Contiguous leads are:
For the precordial leads, it is the neighboring leads (V1-V2 Septal, V3-V4 Anterior, V5-V6 Lateral)
II, III, and AVF are contiguous leads for the inferior portion of the myocardium
I and AVL are contiguous leads for the high lateral portion of the heart (this one is often forgotten)
For the posterior leads, V7, V8 and V9 are contiguous leads
“In those patients with conduction disturbances including bundle branch blocks the Fourth Definition of MI states, “Ischemic symptoms, and presumed new LBBB or right bundle branch block (RBBB) that is not rate-related, are associated with an adverse prognosis. In patients with LBBB, ST-segment elevation ≥1 mm concordant with the QRS complex in any lead may be an indicator of acute myocardial ischemia. Similar findings can be useful in detecting ECG evidence for acute myocardial ischemia in patients with right ventricular paced rhythm.”
I find it best to simply look up the Sgarbossa criteria or the Smith-Modified Sgarbossa Criteria in any patient with a LBBB or pacer:
Concordant ST elevation ≥ 1 mm in ≥ 1 lead
Concordant ST depression ≥ 1 mm in ≥ 1 lead of V1-V3
Proportionally excessive discordant STE in ≥ 1 lead anywhere with ≥ 1 mm STE, as defined by ≥ 25% of the depth of the preceding S-wave"
That does not account for a new RBBB, so if this is ever the case, an old ECG showing RBBB is essential to document or consider consulting cardiology if there is a new RBBB.
For more review, here are some “classic” STEMI patterns from Life in the Fastlane:
Other “classic” patterns that you should know include:
“Tombstone T-waves”
ST-depression
New T wave inversions
Other important ECG changes to know:
Anterior-inferior STEMI due to occlusion of a “wraparound” LAD. This presents with simultaneous ST elevation in the precordial and inferior leads, due to occlusion of a variant (“type III”) LAD that wraps around the cardiac apex to supply both the anterior and inferior walls of the left ventricle
Left main coronary artery stenosis: widespread ST depression with ST elevation in aVR ≥ V1
Wellens syndrome: deep precordial T wave inversions or biphasic T waves in V2-3, indicating critical proximal LAD stenosis (a warning sign of imminent anterior infarction)
De Winter T waves: upsloping ST depression with symmetrically peaked T waves in the precordial leads; a “STEMI equivalent” indicating acute LAD occlusion.
What I document (to remind myself of possible ischemic patterns):
*** rhythm, *** rate. PR interval *** ms, QRS *** ms, and QTc *** ms. *** ST-segment elevation or hyper-acute T waves. *** new ST depression. There were *** T wave inversions compared to prior EKG.
Posterior MI considered with no ST depression in V1-V3. ECG examined for Wellen syndrome - no deep precordial T wave inversions or biphasic T waves in V2-3 and De Winter T waves - no upsloping ST depression with peaked T waves in precordial leads. Sgarbossa criteria considered if LBBB/paced ECG. A large S wave in lead I, a Q wave in lead III, and an inverted T wave in lead III seen in acute right heart strain *** seen. Electrical alternans suspicious for large pericardial effusion *** present.
Independently viewed by attending MD and read by me at the bedside.
The second paragraph acts as a reminder of important patterns that could suggest ischemia. Many times I delete this entirely, but it is much faster for me to quickly review this text and delete it, than to go look up.
On to the Non-ST-elevation MI
<>
On to the Non-ST-elevation MI <>
CC: CP - the NSTEMI
NSTEMI
The non-ST segment elevation myocardial infarction
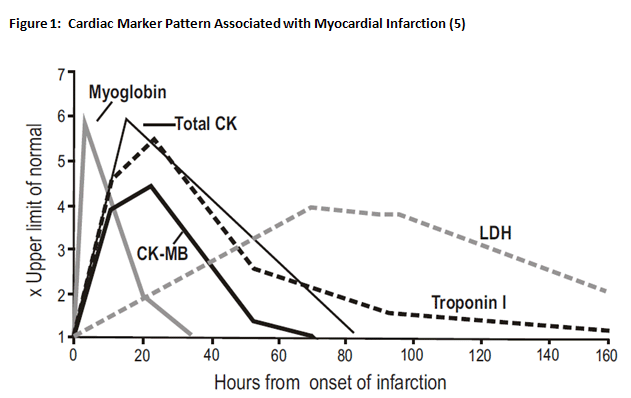
Back in MY early days, we were still using total CK and CK-MB and troponin was the new marker - from: Cardiac Troponin I (cTnI) New Generation Cardiac Marker of Choice. Graphical Representation-Levels of Cardiac Markers in AMI. http://www.tulipgroup.com/ Common_New/ Tech_Pubs_PDF/Cardiac.pdf.
Next, we turn to the non-ST elevated MI. By definition, this is a patient with an EKG that does not show ST elevation (hence the "non") but has evidence of cardiac cell death. This is an easy diagnosis nowadays with increasingly sensitive troponin tests, and it makes for another relatively easy admission disposition and treatment, such as…
NSTEMI should be treated with aspirin.
Heparin is the likely next step needed to prevent a thrombotic event. It would be best if you also utilized nitroglycerin as a vasodilator to keep the patient pain (and hopefully ischemia) free. Also, consider a beta-blocker to decrease the heart's workload.
To summarize, NSTEMIs are managed medically with ASA, NTG, Heparin, and Beta-blockers, but don't stop there.
Watch these patients closely. By definition, these are not going to the cath lab immediately, but they certainly could evolve into a cath lab activating STEMI. Also, consider calling your interventional cardiologist for any hemodynamically stable patient or even a patient whom you cannot control their symptoms (i.e. intractable pain/ischemia) despite maximum medical management. Unstable ventricular arrhythmias (v-tach) are definitely a cue to get on the phone with the cath lab.
Interestingly, I assume there is infarction after seeing a single elevated troponin, but the true definition of myocardial infarction is a 20% serial rise in troponin (at least according to prior standards). I am more familiar with Troponin I which should be repeated in 3 to 6 hours, but I have also worked at ERs that use 1-hour protocols with high-sensitivity troponin. Follow your hospital’s protocol.
Bottom Line: Follow your department’s protocol for utilizing troponin. It is unlikely that you will be making this decision on your own. Also, don’t lose the forest for the trees when an elevated troponin is found. There are other causes including myocardial hypoperfusion might not be ACS but a sick septic or anemic patient.
Despite these nuances, once that troponin is positive and a patient is placed in the NSTEMI bucket, the work-up and treatment is fairly straight forward. In my experience, the difficulties with CP arise with those patients who do not have any EKG or troponin abnormalities, and this is actually the majority of patients who come to the ER with chest pain!
But what about other causes of Chest Pain
???
But what about other causes of Chest Pain ???
CC: CP - the Don’t miss Diagnoses
These are the immediate life-threatening diagnoses that you simply cannot miss. Thankfully, many times these are not subtle presentations, often recognized by nurses and paramedics prior to your evaluation.
According to UpToDate here are the acute Life-threatening conditions you should consider on EVERY CP patient.
Acute coronary syndrome
Acute aortic dissection
Pulmonary embolism
Mediastinitis
Tension pneumothorax
Pericardial tamponade
Let’s address each of these, with a slight change to the last 2 diagnoses on the list. While a small pneumothorax or pericardial effusion is not an immediate life-threatening condition, these are not diagnoses that you want to miss, due to the fact that these can worsen and result in mechanical compression of the heart.
Thus, our list of don’t-miss-diagnoses will include the following:
Acute coronary syndrome (ACS)
Acute aortic dissection
Pulmonary embolism (PE)
Mediastinitis
Pneumothorax
Pericardial effusion
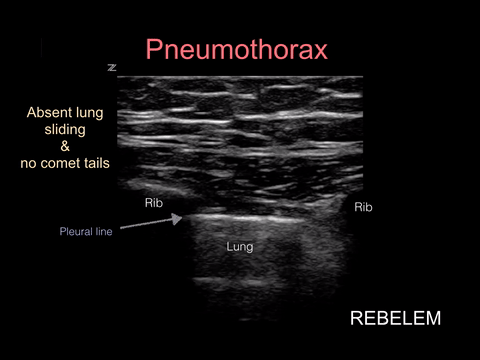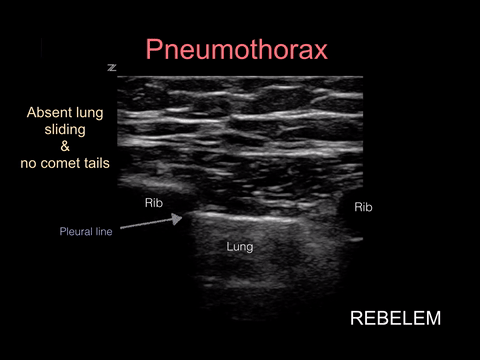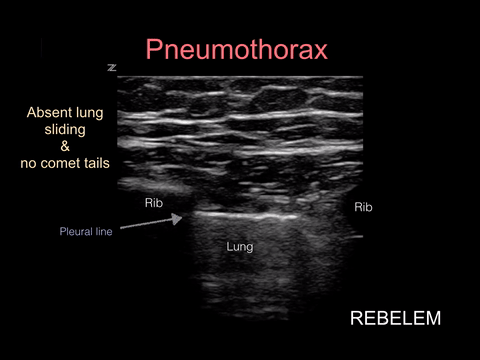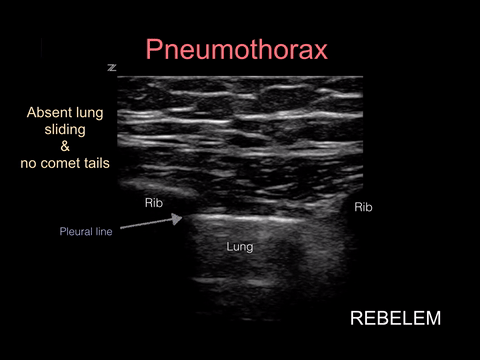
Let’s start with the easiest diagnosis to rule out early —> PNEUMOTHORAX
Don’t wait to get a CXR on a patient with a tension pneumothorax!
Pneumothorax
A tension pneumothorax is most often the result of thoracic trauma or aggressive positive pressure ventilation and usually doesn’t present insidiously with a chief complaint of "chest pain." Rather these are acutely SOB, hypotensive patients and dying in front of you. A patient with a tension pneumothorax will be in extremis, hypotensive, tachypneic, and possibly cyanotic with exam findings such as a trachea pushing away from the side of increased pressure and absent breath sounds on the side of the pneumothorax.
Treatment for a tension pneumothorax is immediate decompression. Typically, the fastest way to do this is a needle decompression, which is beyond the scope and purpose of these posts but can be reviewed in many places and viewed in this video.
Spontaneous pneumothorax, on the other hand, can present with minimal signs and symptoms. Although these don’t often convert to tension physiology, that doesn’t mean it is not dangerous to be discharged home undiagnosed. I strongly suggest considering pneumothorax in every CP patient who presents AND thankfully, this can be ruled out at the bedside with bedside ultrasound!!!
“Lung sonography has rapidly emerged as a reliable technique in the evaluation of various thoracic diseases. One important, well-established application is the diagnosis of a pneumothorax. Prompt and accurate diagnosis of a pneumothorax in the management of a critical patient can prevent the progression into a life-threatening situation. ”
More on this later in our discussion on how to incorporate point-of-care-ultrasound (POCUS) into your physical exam, but in short look for lung sliding at the pleural line and evidence of lung below the pleural line such as A-lines or B-lines. Learn more at https://rebelem.com/ultrasound-detection-pneumothorax/ and https://www.pocus.org/pneumothorax-can-pocus-lung-help/.
Bottom line: You can easily diagnose pneumothorax on bedside ultrasound.
On to —> The THORACIC AORTIC DISSECTION
A thoracic aortic dissection (TAD) might be a bit trickier than a pneumothorax. Though rare, it has high mortality. Classically, patients are supposed to have pain radiating to the back with a tearing or ripping sensation. Hopefully, there is a pulse or BP deficit from one arm to the other that can clinch the diagnosis of ripping off an innominate vessel, but this is dependent on where the dissection goes and is not always present. Other vessels branching off of the aorta that are sheared off by the dissection such as the carotids can cause stroke-like symptoms.
Thus, you must always remember —> CP + neurologic symptoms can = very bad diagnoses such as aortic dissection. Don’t forget to consider this in those patients, but as stated previously, a dissection may not shear off carotid or upper extremity vessels.
It is also essential to consider this in every ST-elevation myocardial infarction (STEMI) patient. A proximal TAD can shear off one or more of the coronary arteries that originate from the base of the arch. A non-perfusing coronary will, of course, cause a STEMI, but not the kind of STEMI we usually think of when a plaque ruptures and clot forms. Imagine if you could pick this up before a patient goes for coronary reperfusion when they really need aortic surgery.
Key takeaways:
Aortic dissections are rare but extremely dangerous.
CP + neuro deficit or radial pulse deficit = dissection until proven otherwise.
Lack of “classic” symptoms does NOT rule out a dissection.
Not every TAD has a widened mediastinum on chest x-ray (CXR)…
..wait, what??? I thought that’s why we got CXR?
CXR
-
Can't r/o dissection
CXR - Can't r/o dissection
More review at http://circ.ahajournals.org/
As you may have learned, thoracic dissections come in 2 flavors, which are useful in determining treatment, but not in making the diagnosis. The purpose of this discussion is to ensure that we never miss this diagnosis in patients presenting with CP. You simply must include thoracic aortic dissection in your differential diagnosis.
Like pneumothorax, the diagnosis may be very obvious from the clues discussed above; however, not every patient with this rare diagnosis has these findings. Thus, we must remember ALL the risk factors, signs, and symptoms of the disease.
High-risk symptoms of thoracic aortic dissection include...
Abrupt onset
Severe pain
Ripping or tearing sensation
High-risk patients are those with...
Connective tissue disease such as Marfan syndrome
Family history of aortic disease
Known aortic valve disease
Recent aorta surgery
Known aortic aneurysm
Finally, high-risk clues are..
Perfusion deficit - pulse difference, systolic BP differential in extremities, focal neurological deficit
Murmur of aortic insufficiency (yeah I would have to look that up too), but remember there can be a wide pulse pressure with aortic insufficiency as well (look up pulse pressure if you need to)
Hypotension/shock
With all these risk factors, is there at least a good clinical decision tool? Um, sort of. The Aortic Dissection Detection Risk Score (ADD-RS) is externally validated but not routinely used, in my experience. Not that I don’t find it helpful, as it does a great job of reminding me of all those high-risk features above.
The ADD-RS + D-dimer (the ADvISED study algorithm) attempted to combine the ADD-RS score with a d-dimer to rule out aortic dissection, but unfortunately has not been externally validated. This article from the Annals of Emergency Medicine 2015 also tried to use a D-dimer of >500 ng/mL as a cut-off to rule out TAD, and although it was very sensitive (98%), this was just not high enough for such a dangerous disease.
“Level C recommendation - In adult patients with suspected nontraumatic thoracic aortic dissection, do not rely on D-dimer alone to exclude the diagnosis of aortic dissection.”
I suppose if the patient had no major risk factors or features (see lists above) AND had a normal CXR and bilateral blood pressures AND you had a low suspicion (i.e. low pre-test probability), you could THINK about buffing your chart by getting a D-dimer <500. The problem with this approach is, are you prepared to CT aortogram all the patients with a high D-dimer???
Bottom line: You can't treat what you don't diagnose. Using the Aortic Dissection Detection Risk Score (ADD-RS) is a good start. It can remind you of important risk factors and disease features.
On to —> PULMONARY EMBOLISM
For obvious reasons, pulmonary emboli (PE) are included on the “don’t miss” list, and similar to aortic dissection, they can present along a spectrum from obvious to obscure. A massive PE will likely cause the patient to be hypoxic, tachycardic, and hypotensive (see official criteria in this article in Circulation). Severe right heart strain will lead to jugular venous distention (JVD) and the lungs will be mysteriously clear (unlike left heart failure). A massive PE is an immediate life threat, and you need to make the diagnosis early, just like a tension pneumothorax and aortic dissection.
Again, you could do a bedside echocardiogram early to look for right heart strain (more on this later). An unstable, hypotensive patient may not be stable enough to go for a CT to confirm the diagnosis. Learning to recognize right heart strain on bedside echo is not easy, but you don’t have to rely on just the images. Looking at the whole clinical picture, risk factors, symptoms, JVD without pulmonary edema, and other clues can lead you to the diagnosis even without exceptional echo skills, but when you combine your clinical suspicion AND rule out other causes that could lead to this patient being unstable - tension pneumothorax (discussed above) and pericardial effusion with tamponade (discussed below) - you can be much more confident in the diagnosis even without recognizing right heart strain.
An excellent discussion of right heart strain on echo can be found here - https://coreultrasound.com/5msblog-rhs-1/
More valuable teaching on identifying RV strain at the bedside can be found here - https://rebelem.com/diagnosis-right-ventricular-strain-transthoracic-echocardiography/
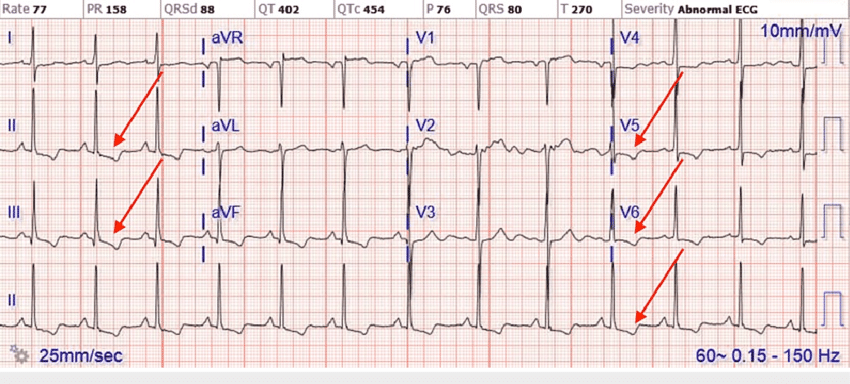
Speaking of right heart strain, an EKG is a quick and easy test that can also help your differential diagnosis. The most common EKG finding in PE is tachycardia, which is not all that helpful, but you also may have learned the “classic” SI QIII TIII pattern – deep S wave in lead I, Q wave in III, inverted T wave in III . Unfortunately, this “classic” pattern is only found in 20% of patients and isn’t sensitive or specific enough to be all that useful. Right ventricular strain and right axis deviation are also good to look for but don’t confirm the diagnosis completely (click here for more ECG findings from LITLF).
This EKG shows: RBBB, Extreme right axis deviation (+180 degrees), S1 Q3 T3, T-wave inversions in V1-4 and lead III, and Clockwise rotation with persistent S wave in V6 according to the smart authors at https://litfl.com/ecg-changes-in-pulmonary-embolism/.
Acute right ventricular dilatation due to massive PE - https://litfl.com/right-ventricular-strain-ecg-library/
Hopefully, this review helps clue you into all the signs and symptoms of a massive PE that you can assess right from the bedside, even before confirming with a CT pulmonary angiogram (CTPA), if the patient is stable enough to get one. Many of these may also be present on submassive PEs, and you should have a low threshold to get a CTPA on these more stable patients.
The difficulty in diagnosing PE lies in the patients who are not critically ill or only a few risk factors or signs that point toward PE as the #1 most likely diagnosis. These are usually patients who have smaller, sub-segmental PEs that we still cannot miss, due to the danger that they propagate and kill the patient. Therefore, we must be careful not to miss any PEs, no matter the size or presenting symptoms.
Thankfully, unlike aortic dissection, there are externally validated, well-known, and broadly accepted risk stratification tools that we can use in any patient with any chance of having a PE - which in terms of the undiagnosed CP patient, is every one of them without a confirmed diagnosis. Of course, we don’t have to use these tools in patients with a high risk of PE, but then again, who are those patients?
My advice is to apply the following pathway to every patient with symptoms that could be caused by a PE, unless you already have a confirmed cause of those symptoms (and even then, unfortunately, patients can have pneumonia and a PE).
What is your gestalt? After obtaining a history and performing a physical exam (that can include bedside echo), is PE the most likely diagnosis? If so, go straight to CTPA.
If you are unsure, or inexperienced, apply the Well’s Criteria for PE. Click the link or see the image below.
Any moderate or high-risk patients should get a CTPA.
If low risk, move on to step 3.
If low risk by Well’s Criteria for PE, apply the PERC score.
If any criteria are met on the PERC score, the patient needs further work-up; go to step 4.
If the PERC score is zero (and your pre-test probability of PE was < 15%, see image below), the chance of PE is < 2%, and no further work-up is needed.*
Obtain and interpret a D-dimer.
Your lab will likely have a pre-set cut-off for a “positive” result, usually 500 ng/mL. Below this is considered negative, and no further work-up for PE is needed.**
If the D-dimer is >500, then you can consider applying the Age-adjusted D-dimer cutoffs.
If above cutoff, proceed to CT PA.
If below the age-adjusted cutoff (10 x patient’s age), then the previously low-risk patient by Well’s Criteria, is almost zero risk for PE, and further work-up not necessary.
*In most cases, this effectively rules out PE or is not high enough to outweigh the potential risks of a CT scan (radiation exposure), but I suggest that you consider documenting this < 2% risk and even talk to your patient in a shared decision-making manner to discuss obtaining a CT.
**I also suggest discussing this with your patients, especially when the result is close to 500. I have seen D-dimer tests at 490 and 505 on separate occasions. I have told these patients that there is good evidence to support 500 as a cut-off, but this is not a magical number, and I have used shared decision-making in these cases.
For more information go to MDcalc.com and Well’s Criteria for PE.
For more information go to MDcalc.com and PERC score.
My pathway using Well’s criteria, PERC, and age-adjusted D-dimer is a bit wordy, but it is worth the read so that you can apply this to your medical decision-making (MDM).
Final Caveat - This pathway does not include pregnant patients who cannot be classified as low risk. The YEARS Algorithm can be helpful in these situations.
Bottom line: CT every patient at moderate or high risk for PE, and document your medical decision-making on every patient at low risk for PE with no alternative explanation of their symptoms. Feel free to use my pathway above as a template for your own MDM.
This is, in my opinion, the easiest bedside echo to interpret with huge ramifications if found. In fact, I'm so keen to find this diagnosis early, that I see value in teaching paramedics to pick it up in the field. See my review article, The Future of Ultrasound in Prehospital Resuscitation.
Finally —> Pericardial Effusion with Tamponade
The last of the immediate life threats to consider is cardiac tamponade, although, chest pain is not likely going to be the only presenting complaint. A patient with a pericardial effusion large enough to significantly disrupt LV function is going to be hypotensive. By definition, cardiac tamponade occurs when the amount of fluid in the pericardial sac compresses the ventricles and leads to a significant decrease in cardiac output and shock.
There are several causes of pericardial fluid accumulation, including blood from blunt or penetrating trauma which occurs with trauma. Cancer can also cause a pericardial effusion which usually slowly accumulates. These patients are more likely to present with signs and symptoms of hypotension, rather than just chest pain.
In terms of patients presenting with chest pain, pericarditis is the diagnosis that is important to catch. While this is a rare cause of pericardial tamponade, it is an important diagnosis to include in your differential.
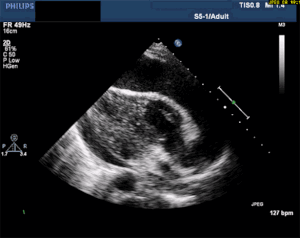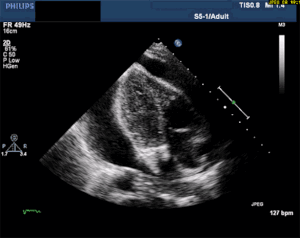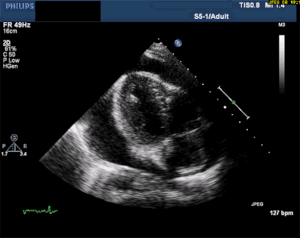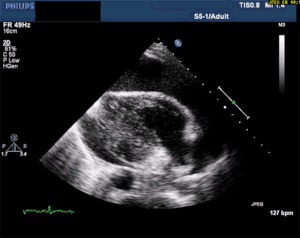
Thankfully, a large pericardial effusion is the easiest diagnosis to make on bedside ultrasound! Just place the probe on the chest, and if you can get a view of the heart, you can see the black effusion surrounding it. Additionally, the lack of large effusion on bedside echo can rule tamponade out. Either way, bedside echo is your friend.
Bedside Ultrasound - for EVERY CP patient
As you can see from multiple images above, early ultrasound can help identify these life-threatening conditions. Early imaging by performing a bedside echocardiogram upon initial evaluation can rule-in or rule-out some of these life threats with fairly high accuracy. In fact, there are several protocols for pre-hospital point of care ultrasound (POCUS) that have varying success rates reviewed in this paper:
The Evolving Role of Ultrasound in Prehospital and Emergency Medicine - ClinicalKey
The absolute best way to improve your sonographic skills is to practice, practice, practice every time you see a patient with chest pain, even when you have a very low suspicion of finding anything on your exam. At best you will pick up surprising findings, at worst you will see many, many normal hearts while honing your technique.
What to document:
Dangerous causes of CP were considered with no evidence of pneumothorax on bedside exam and CXR.
The patient has *** risk factors for aortic dissection and symptoms were *** consistent with dissection. The Aortic Dissection Detection Risk Score (ADD-RS) was reviewed; *** further work-up was pursued.
The patient *** is hemodynamically stable with *** exam findings to suggest profound right heart failure (hypotension, clear lungs, JVD) that could suggest massive PE or pericardial tamponade. A bedside echo revealed *** pericardial effusion.
Sub-massive PE was also considered. The patient was *** risk by Well’s Criteria for PE. If low-risk, the PERC score was ***. Further evaluation with D-dimer is *** warranted. If the D-dimer is >500, then considered applying the Age-adjusted D-dimer cutoffs. The YEARS Algorithm was used in pregnant patients.
I’ll admit, that documentation above was a bit wordy, which is why I’m open to any of your suggestions. Thanks!
You did it!!! You considered life-threatening causes of CP.
But what about other causes of Chest Pain
???
But what about other causes of Chest Pain ???
CC: CP - Undifferentiated
But what if the chest pain (CP) isn’t myocardial infarction???
Thus far we have reviewed the STEMI and NSTEMI, each of which both have clear EKG findings and elevated markers of myocardial injury, usually troponin. There are also many other causes of “chest pain” with very obvious findings, like a stab wound to the chest. I have seen several times, herpes zoster on the chest wall triaged as “chest pain” in patients with communication impairments like dementia.
In textbooks, there are huge lists of potential causes of chest pain that attempt to be exhaustive.
Fairly exhaustive list, but somewhat impractical to clinical application with causes such as trauma and food impaction not likely to be missed with an actual history and exam.
I don’t find these huge lists helpful clinically. While CP may be part of the presenting complaint, many of the diagnoses above have other obvious features, such as trauma or excessive vomiting. Rib fractures and esophageal rupture usually have historical clues that do not obscure the diagnosis. Similarly, without trauma or rash several diagnoses below can be excluded.
In other words, you don’t need an exhaustive list such as this in every CP case, as many times there will be an obvious cause.
Instead, I would like to focus on…
UN-differentiated CP.
That is to say, there is no OBVIOUS cause of CP on the initial history, exam, EKG, and labs (all of which we will get to separately).
It is the "undifferentiated" patient that takes careful thought and documentation before discharging home. Anyone can admit a patient with undifferentiated CP for further work-up, but this is becoming increasingly rare with widespread use of accelerated diagnostic protocols (like the HEART pathway discussed later) and higher sensitivity troponins. The danger here is not in missing an MI, but in only looking for an MI and missing other important diagnoses.
“It is forgivable to miss a rare, devastating diagnosis. It is not forgivable to not think about, and document, it.”
This is why I so strongly believe in creating templates for your patient care. I use them as a reminder to consider potential dangerous causes of a patient’s presenting symptoms, and a shortcut in my documentation, having already laid out the rationale for testing or not testing. I also find it much faster to erase irrelevant diagnostic considerations, such as in the case of CP proven to be an MI by a positive troponin, than to dictate into my documentation all potential dangerous causes in a patient I am planning to discharge home.
Back to the matter at hand, however, and let's discuss ways to create a template or smart phrase to easily import into your notes of every undifferentiated CP patient to remind you of dangerous dx to consider and speed up your documentation to discharge. What conditions should be included in your MDM? Think about it for a second and try to list 10 diagnoses that cause "non-obvious" CP including all life-threatening causes.
(I'll wait)
What did you come up with? According to UpToDate here are the acute Life-threatening conditions that you should consider on EVERY CP patient.
Acute coronary syndrome
Acute aortic dissection
Pulmonary embolism
Tension pneumothorax
Pericardial tamponade
Mediastinitis
Thankfully, some of these have fairly obvious presenting features making them hard to miss. Yet, as we can see from litigation data, sometimes immediate life-threats are missed. I suggest considering these every CP visit.
From the list above, I don't consider the last 3 to be in the "undifferentiated" CP category because these patients are likely ill-appearing. It is debatable if they even need to be included in my MDM of patients that I discharge, but after reflection, I think they are worth mentioning as considered but not pursued because they are of minimal suspicion due to lack of risk factors or exam findings.
Of course, there are other possible causes of CP that are not immediate life threats and are listed here...
Cardiac causes:
An infection/inflammation process like pericarditis should be considered if symptoms support it.
Myocarditis would be accompanied by troponin elevation.
Endocarditis usually has other stigmata of septic emboli on examination.
Valvular disorder such as MVP could cause a brief CP sensation and aortic stenosis might cause chest discomfort (possibly through demand ischemia) with acute severe obstruction or aortic or mitral regurgitation which would likely cause pulmonary edema and poor perfusion.
Pulmonary cause:
Infections (pneumonia, tracheitis, and bronchitis) should have accompanying symptoms like fever, cough, etc.
Chest tightness is a common complaint with asthma exacerbations.
Pulmonary malignancy can cause chest pain particularly if there is pleural involvement. Chest heaviness or discomfort may be noted with pleural effusions
GI causes:
Gastroesophageal reflux and esophageal spasm, rupture (Boerhaave syndrome), or inflammation can all present as chest discomfort.
A sliding hiatal hernia may result in chest pain. Pain from pancreatitis can be referred to the chest.
MSK:
Rib contusions and fractures would have a history of trauma
Intercostal muscle strains probably would have an inciting event like coughing
Costochondritis should be associated with chest pain with palpation, but this should not be used to exclude other diagnoses, only to support a suspicion of MSK etiology.
Derm: herpes zoster, cellulitis, abscess, and dermatitis all have obvious skin findings (usually in a patient that can't communicate the nature of pain)
Depending on your experience and preference, you may or may not want to include reminders in your own CP template to look for signs and symptoms of the above diagnoses, but we’ll get to that…
On to..
the don't miss Dx!!!
On to.. the don't miss Dx!!!
CC: CP - other important Dx to Document
Other stuff that causes chest pain...
So there you have it. Don't walk into or out of a room with a patient with chest pain without thinking of MI, massive PE, pneumothorax, and dissection.
Next up, all of the other dangerous and not-so-dangerous causes. To name a few...
Pericarditis/myocarditis
Pneumonia/pneumonitis
Esophageal rupture - Boehaaves syndrome
Esophageal spasm
GERD
Incarcerated diaphragmatic hernia (huh? Yeah, that's a thing)
Costochondritis aka Tietz syndrome
Pyelonephritis (I've never seen this)
Herpes zoster (obvious once rash shows up)
Idiopathic (we’ll never know)
Lists like the one above, and attempting to diagnose based on location and radiation is a waste of time. I highly doubt any trained clinician is going to miss a ruptured esophagus!!! A good history and examination will usually elucidate a non-intrathoracic cause of chest pain. For example,
If a patient is complaining of CP and vomiting blood, think Mallory-Weiss tears, or
If there is a dermatomal rash on the chest, think of shingles, or
If the chest is tender, it could be costochondritis.
The list of causes for (non-obvious) undifferentiated CP is long and can include the following:
Angina (without infarction)
Endocarditis
Acute chest syndrome (in sickle cell patient)
Sub-massive PE
Pericarditis
Pneumonia
Aspiration pneumonitis
Pancreatitis
GERD
Very rare causes
Mediastinitis
Perforated peptic ulcer
Spontaneous coronary artery dissection
Stress (takotsubo) cardiomyopathy
It is hard to say what from the list above should be routinely included in your MDM, but here is what I have come up with (for now)…
Infectious etiology considered. History and lung exam do *** support pneumonia or respiratory infection. Patient is not at risk for aspiration pneumonia. There is *** history of recent IVDU and *** signs or septic emboli on examination to suggest endocarditis.
Gastritis, GERD, and pancreatitis were considered with *** risk factors or historical clues to support this.
Pain is *** increased with lying or relieved with sitting up to suggest pericarditis with *** pericardial effusion seen on bedside ultrasound.
Clinically significant PE was also considered with the patient having *** risk factors. Well's criteria was used to risk stratify with the patient *** for PE. The PERC criteria was *** applied. Further work-up *** indicated.
What about anxiety and chest pain?
Well, it turns out a lot of people in the ED with chest pain (CP) have anxiety about it. That seems to make sense because the reason they came to the ED was because they were worried about the CP. Many people will actually ask me, “Do you think that this pain could be because of my anxiety (or stress)?” Usually, my response is something along the lines of “I wish I had a test for that.”
Anxiety and other emotions can cause many symptoms and physiologic changes (see my Takotsubo syndrome case report). In addition, this study, no surprise, found that about 50% of low-risk CP patients presented during a time of high anxiety. Furthermore, many of these types of patients end up re-presented at a later time. Now, I think it is silly and foolish to make a diagnosis of anxiety “causing CP,” but that does not mean that it isn’t work talking to the patients about their anxiety. At minimum, this might put the patient’s mind at ease (perhaps decrease anxiety), and it may decrease repeat visits. When I do this I generally share the statistics for the HEART score or even show them the calculator and their risk of 30 day adverse outcome. Again, I don’t label CP as “cause by anxiety" but that anxiety can be a strong component of presenting symptoms.
Should you document a discussion about anxiety with patients with no apparent cause for CP?
I don’t think that you will find a consensus opinion, but I would love to hear your thoughts. Personally, I often talk to patients about anxiety when there is no cause found for their CP, but it depends mostly on the state of the patient and if think that they would be open to a discussion. A line I have often told patient’s is that I always take a complaint of chest pain seriously and don’t consider anxiety alone as the cause until I have considered dangerous diagnoses. Sometimes my work-up and consideration for dangerous causes of CP can be done right at the bedside with a good history and exam. I can listen for pneumonia or pneumothorax. I will check vitals signs (as you will see) and look at an ECG and labs if necessary.
With anxious patients, I have found it helpful to list out diagnoses that I know are NOT present in terms that they can understand, such as…
”I do not see signs of a heart attack on your EKG or lab work-up and you don’t have any risk factor or signs for blood clots in your lungs. Your symptoms and exam don’t sound like a collapsed lung or pneumonia. Are there other things that you might be worried about that I could address?”
Many patients and family members appreciate a speech like that so consider adding it to your “standard” work-up if you don’t already. As for routine documentation, because this conversation varies so much from patient to patient, I don’t include it in my CP template.
On to the vitals and exam!!
What can the vitals tell us
???
What can the vitals tell us ???
CC: CP - the VS
Vital Signs are Vital! Don’t skip.
Just as you will likely be able to see an EKG before you interview and examine your CP patient, you will also probably have a set of VS from triage, but what clues can you gather from the VS to tailor your H&P? Here are just a few things to think about as you review the VS.
Temp -
Fever? Look for infection - pneumonia, myocarditis, endocarditis
Low temp - don’t miss sepsis
HR -
Tachycardia? PE, almost anything else but think twice about d/c when tachycardia
Bradycardic? Perhaps well-conditioned patient - correlate with BP
BP -
BP high? Both systolic and diastolic high - -dissection risk.
Hypotensive? Again, sepsis high on the list. Sudden valvular disease can also be considered, as in the example below…
Notice anything weird about the vital signs in the image, other than the monitor being way too high? Yes, the patient is technically bradycardia, but the systolic BP looks okay so probably not that clinically significant. No, don’t look at the ECG tracing, that’s not part of the VS. Yeah, I don’t know what the blue 13 or green 14 represent, but let’s just say respiratory rate. The 99 is probably the pulse oximetry; looks good.
The number that sticks out to me is the diastolic BP of 51. Why is that weird? I’ll give you a minute to think.
The patient above has a pulse pressure of 72 (SBP - DBP = PP). While technically not “widened” by the strict definition, the low diastolic BP could be a clue about peripheral vascular resistance being low. This could be normal, or it could be an early sign of sepsis that could lead to shock. With the HR not elevated in this particular example, I am not as concerned.
Nevertheless, even “normal” VS can offer clues that may be obvious in retrospect, such as the patient progressing from systemic infection to septic shock. Based on my experience with a patient with acute aortic valve insufficiency, the vital signs were a “vital” clue that I missed early in the work-up. High-output heart failure can also be a puzzling initial presentation in the a patient with “good” BP yet showing CHF.
Another, real life example of a widened pulse pressure being a clue to cause of a patient’s symptoms was a case of aortic insufficiency I almost missed on a patient presenting with “flu-like symptoms” who had endocarditis with destruction of his aortic valve. Check out the image below for more on wide pulse pressures.
Bottom line:
Be wary of abnormal vital signs!!!
Consider charting the following or including it in a template to force yourself to look at the auto-populated VS every time.
Vital signs reviewed with *** abnormalities.
CC: CP - the Hx
First, does a history even help in determining the cause of undifferentiated CP?
By definition we are talking about undifferentiated CP, not CP caused by something obvious the patient told you, like “My chest hurts ever since it hit the steering wheel in a car crash.”
Remember OPQRST? Or SOCRATES?
I can’t, and I don’t know if “Did you call a lawyer?” fits in either of those, but I have heard patients say that their lawyer told them to go to the ER several days after an MVC, and it was quite informative.
But does the Hx matter in undifferentiated CP???
Of course it does!
Salim Rezaie, "Chest Pain: What is the Value of a Good History?", REBEL EM blog, November 1, 2013. Available at: https://rebelem.com/chest-pain-value-good-history/.
Because we are talking about undifferentiated CP, I am not going to include EVERYTHING a patient might say that could be a clue to the etiology of their symptoms. That list would be quite long and include things like “Do you have a knife in your chest?”
Narrowing in on identifying acute coronary syndrome (ACS), research has found there are some specific characteristics of the “HPI” that can increase or decrease the likelihood that a patient has ACS and/or AMI.
A history that includes CP PLUS radiation, diaphoresis, N/V, and/or exertion INCREASES the likelihood of ACS. Therefore, it is a great practice to include all these features in a thorough HPI.
Including the presence of pleuritic, positional, stabbing, or palpable pain, can decrease the likelihood of ACS in your routine HPI is also advisable as a way to prompt you to consider pericarditis, PE, GERD, pneumonia, MSK, or other etiologies
Please read the full article for context and remember that increasing or decreasing the likelihood of CP caused by ACS does NOT rule out the presence of AMI. Instead, you can use these clues to help you think of potential OTHER causes of CP.
“In the event of a missed diagnosis of myocardial infarction, thorough documentation of the absences of features most common in ACS is important. ”
But don’t stop there!!! Comprehensive documentation includes consideration for ALL potential dangerous or critical causes of CP.
The following list includes the Dx - followed by the pertinent hx and risk factors for many diseases that could present with CP. These can broadly be divided into cardiac ischemia and/or inflammation, pulmonary etiology, vascular, thoracic wall, and extra-thoracic etiologies
Cardiac ischemia/inflammation
ACS - CP with exertion, CP plus diaphoresis, similar to prior angina/MI, hx of HTN, DM, PAD, CAD.
Takotsubo cardiomyopathy - mimics ACS, sudden onset, emotional distress
Coronary vasospasm - cocaine use, family history
Pericarditis - CP worse lying, better sitting up, pleuritic pain, recent viral infection, prior pericarditis
SCAD - pregnant, connective tissue disease
Myocarditis - recent illness, sx of heart failure
Endocarditis - IVDU, fever, immunosuppressed
Thoracic vascular disease
Aortic dissection - sudden, tearing pain radiating to the back, Marfan or connective tissue disorder, age > 50, hx of HTN and/or PAD.
Thoracic aortic aneurysm - known history (does not usually present as CP)
Pulmonary
PE - pleuritic, hemoptysis, DOE, active cancer or clotting disorder
Pneumonia - fever, cough, pleuritic CP
Asthma/COPD - hx of either, SOB, chest wall pain, worse with deep inspiration
Pneumothorax - sudden, resolved pain, tall-thin, SOB
Mediastinum pathology
Mediastinitis - possible neck/dental source/pathology, Ludwig angina, sx of infection
Esophageal foreign body - occurs with swallowing, regurgitation/unable to swallow
Esophageal rupture - forceful vomiting
Extra-thoracic (abdominal) source
Esophageal/GERD/gastric ulcer - acid taste, occurs before/after eating
Gallbladder disease - post-prandial pain, RUQ pain, radiation to R shoulder
Pancreatitis - pain with eating, EtOH/gallstone associated
The problem that we run into again, is the sheer length of the above list and number of questions, clinical risk factors, and features that would need to be included. Here, I defer to your preferences in your documentation. I believe that it is easier and faster to DELETE unnecessary portions of an HPI that you did NOT ask, than it is to dictate ALL the portions that you did cover. PLUS, a long list of features and risk factors MAY even remind you to ask questions relevant to rare diseases.
Additionally, several commonly used clinical decision making tools use “suspicion,” historical features, and other risk factors to calculate risk scores. These include the HEART pathway, Well’s criteria for PE, and the PERC rule.
Outside of the VS, PE, and lab findings, you must remember to ask patients their history of DM, obesity, HTN, and hyperlipidemia, recent/current smoking status, family hx of CAD <65, prior MI/PCI/CAB, CVA/TIA, or PAD for the HEART pathway. For the PE scoring systems, you should ask about leg swelling, recent surgery or trauma, prior PE or DVT, cancer treatment within 6 months, hemoptysis, and hormone use including oral contraceptives and hormone replacement.
The question is - Can you create a “standard” HPI template that reminds you to ask all those questions?
Possible HPI documentation template:
Patient presents with chest pain for *** described as ***. There is *** radiation to ***. *** diaphoresis with pain. The pain is *** worse with exertion and is *** relieved with rest. There is *** pain with breathing or deep inspiration. The pain is *** with sitting; *** in pain with lying flat. *** recent fever or infectious symptoms. *** cough recently. Pain is *** associated with eating. *** leg or feet swelling.
The patient has *** known CAD. *** prior DVT or PE. There have been *** recent immobilizations or surgeries. *** unilateral leg or calf pain. The patient does *** take exogenous hormones or OCP. The patient *** smokes cigarettes. *** recent cocaine use. *** history of IVDU.
The end of the above HPI also includes the PMH and social history. It depends on your documentation style, if you want to include these in the HPI or elsewhere. Alternatively, you could add reminders to your template that include risk factors within the ROS, PMH, Family History, etc.
Expert tip: While many EHRs automatically import PMH, FH, and other aspects of patient history collected at triage or in prior visits, it is still possible to miss many risk factors that were not specifically asked about in prior visits. For example, a patient may have started taking contraceptive pills or had a recent surgery not automatically imported into the chart. It is best practice to review all information that is used in medical decision-making at the time of the visit, not relying on prior documentation that may not have addressed cocaine or illicit drug use.
Your template or documentation could also be broken up like this…
HISTORY OF PRESENT ILLNESS
Patient presents with chest pain for *** described as ***. There is *** radiation to ***. *** diaphoresis with pain. The pain is *** worse with exertion and is *** relieved with rest. There is *** pain with breathing or deep inspiration. The pain is *** with sitting; *** in pain with lying flat. *** recent fever or infectious symptoms. *** cough recently. Pain is *** associated with eating. *** leg or feet swelling. *** unilateral leg or calf pain.
PAST MEDICAL HISTORY
Reviewed for risk factors of prior CAD or MI, HTN, DM, PE or DVT, or other vasculopathy. *** history of cancer. *** history of VTE.
SURGICAL HISTORY
*** recent surgeries or immobilizations.
CURRENT MEDICATIONS
*** current oral contraceptive pill.
FAMILY HISTORY
*** history of premature heart disease or MI under the age of 55.
SOCIAL HISTORY
Reviewed for current or recent smoking, cocaine use, or IVDU.
REVIEW OF SYSTEMS
General: *** fever.
Neuro: *** recent syncope.
Skin: *** rash. *** lesions on hands or feet.
ENT: *** rhinorrhea or sore throat.
Neck: *** swelling or pain.
GI: *** nausea.
Extremities: *** lower extremity swelling.
Psych: *** anxiety. *** depression.
With recent changes in documentation requirements for coding and billing purposes, the details of your HPI and ROS are no longer necessary for higher-level billing. This means you no longer need to document a 10-point ROS or HPI with 4+ characteristics. That does not mean that you no longer need to collect this information.
To shorten your documentation, you may write “History and physical examination appropriate for complaint” but this does little to defend yourself in court when asked how thorough you were in your H&P during a patient visit several years ago, now being litigated. As I have said before, it is easier to delete what you did not do, than to dictate what you did ask. Beyond that, a template might even remind you to back into a room and ask the patient if they had recently used cocaine which could greatly change the treatment plan.
However, you document, consider both efficiency and accuracy when creating a template that can accurately reflect what you actually said and did during each individual patient visit.
CC: CP - the PE
Ok, so you have a pt with no obvious cause for CP, what do you examine and why? Let’s say you have a known cause for CP, do you need to examine anything?
In a word, YES! Of course, you have to do a physical exam (PE)!!! It can offer SOME useful information.
The problem is…
You’re doing the physical exam wrong!!
Stop doing a head-to-toe PE, and definitely stop documenting a standard normal exam that you didn’t do!
Those changes are congenital, Dr. House!
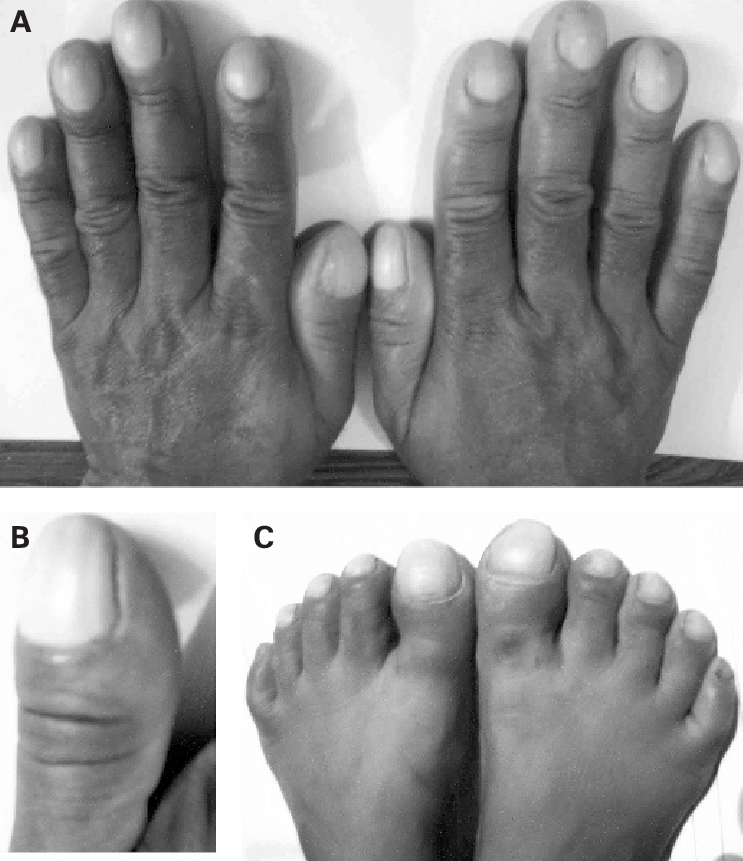
Do you even know what clubbing is? Stop documenting this finding that is either genetic or took decades to occur due to chronic hypoxia. It’s probably not causing CP. Instead, look at the extremities to assess perfusion (cyanosis, cap refill) and for edema that could be part of new heart failure.
You’re a smart clinician (because you’re reading this post), don’t waste your time looking/listening for things that aren’t pertinent, just to make it look like you know what you’re doing. Go down you’re differential diagnosis and look/listen/feel for them!!!
Look at the skin for herpes zoster, abscess, cellulitis. I’ve seen it before on a dementia patient who didn’t need a “CP work-up” for a painful zoster on her chest.
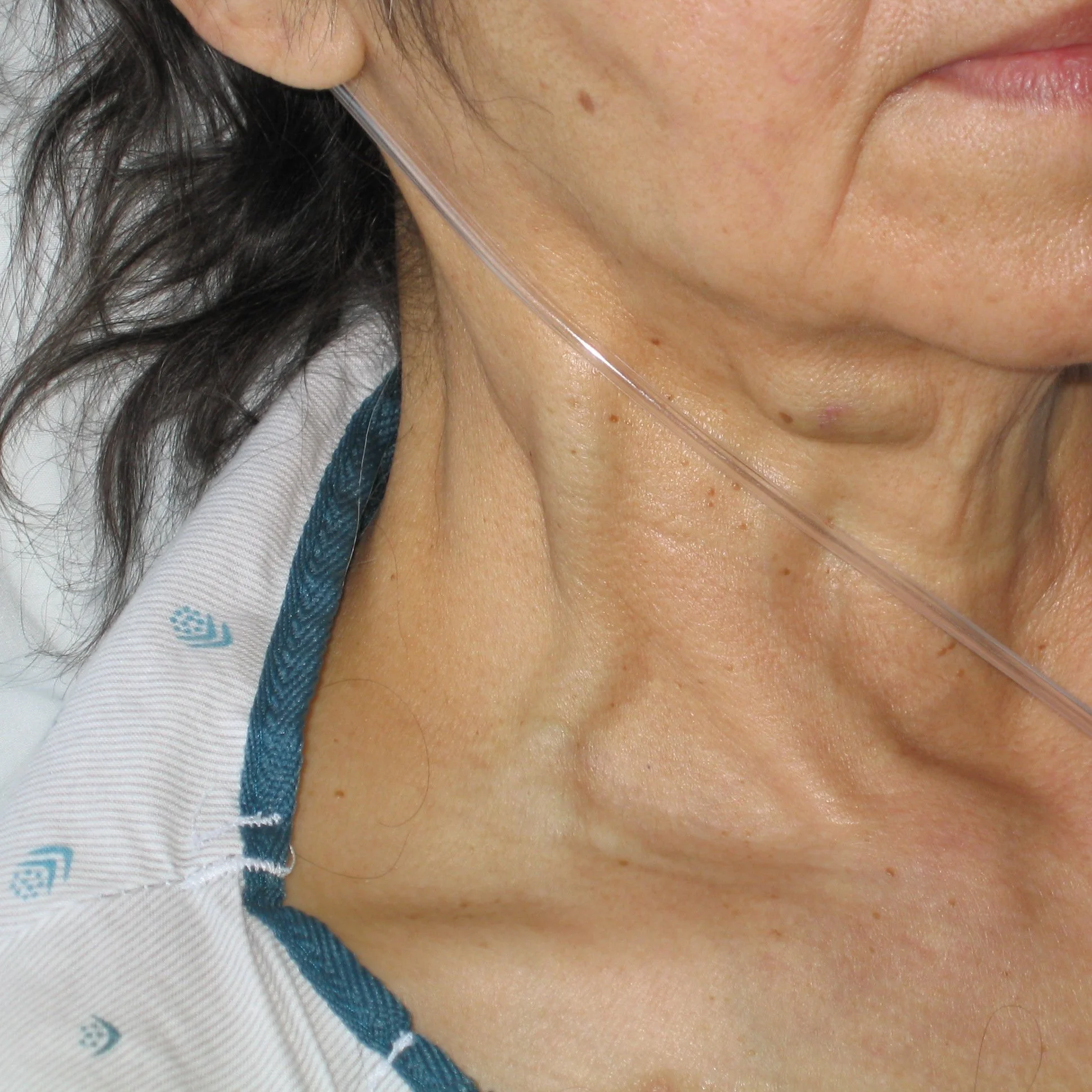
Actually, look at the neck for JVD of heart failure.
The abdomen lies fairly close to the chest and I have been guilty of overlooking acute cholecystitis or other intra-abdominal pathology a time or two only to go back and examine the abdomen and test for a Murphy’s sign.
Don’t miss… A Pneumothorax
Don’t rely on just your stethoscope (and percussion?) to detect a pneumothorax, and DEFINITELY don’t wait for chest x-ray to detect a tension pneumothorax!!!
This patient is dying while waiting for a chest x-ray to be taken!
If only there was a quick, reliable method of detecting pneumothorax at the bedside…oh wait, there is.
Evidence:
Conclusion: The present meta-analysis showed that the diagnostic accuracy of CUS was higher than supine CXR for detection of pneumothorax. It seems that CUS is superior to CXR in detection of pneumothorax, even after adjusting for possible sources of heterogeneity.
Conclusion: The diagnosis of pneumothorax using ultrasound is accurate and reliable; additionally, it can result in timely diagnoses specifically in neonatal pneumothorax. Using this method facilitates the therapy process; lack of ionizing radiation and easy operation are benefits of this imaging technique.
…a few physical exam findings and the clues they may give us to an underlying disease.
Heart sounds - listen for a rub of pericarditis, muffled sounds of pericardial effusion, a new murmur of a valve rupture or regurgitation
Jugular venous distention - might indicate right heart backup in pulmonary embolism, cardiac tamponade or acute heart failure
Lungs sounds - listen for lack of breath sounds in pneumothorax and adventitious sounds like crackles in CHF or rhonchi in pneumonia
Pulses in all 4 extremities - a pulse deficit in any extremity could indicate an aortic dissection
Neuro deficit could also scream dissection (of the carotids) in the context of tearing chest pain.
This is JVD - you can look at your patient’s neck and get clues about what their heart and lungs might sound like!
Use your stethoscope as a “test” to increase or decrease the likelihood of certain diseases.
Thinking of CHF? Start at the bases and listen for rales.
Focal rhonchi occurs in lobar pneumonia so spend the time listening to every lobe if that’s on your differential.
If you think pneumothorax, just go right to POCUS (my opinion), or listen specifically for decreased breath sounds.
Do you know how many times I’ve see RRR in a pt with a-fib? Or no murmurs on a pt with aortic stenosis? It’s better to chart nothing. Instead, auscultate for specific findings pertinent to a differential Dx of CP.
What might that be, you ask?
Systolic ejection murmur heard best at the base of a pt with no know history of aortic stenosis or a loud AS murmur in a pt whom you’re concerned now has critical stenosis causing heart strain.
Aortic insufficiency would sound like a diastolic murmur in the context of a widened pulse pressure you already found.
Listen carefully over the mitral and tricuspid valves susceptible to vegetation in endocarditis.
Listen for that pericardial rub of pericarditis? Yeah right. Muffled heart sounds??? Just grab the ultrasound and look for a pericardial effusion.
Finally, if you have a pt who goes into flash pulmonary edema after an MI, check for a systolic murmur at the apex from acute mitral regurgitation from papillary muscle infarction.
If you didn’t follow any of that, consider retiring your stethoscope or at least relearning how to use it. Despite being a nearly outdated tool, it can offer some specific information.
Let’s recap…
The PE is a useful part of the evaluation of a pt with undifferentiated CP.
Don’t go through the motions of a PE. Take that time to assess for actual diseases.
The list of pertinent disease that can present as undifferntiated CP include the following:
Herpes Zoster or other skin pathology
Pneumothorax - heaven forbid you wait until you get a CXR to consider this!
Pneumonia that in retrospect was fairly obvious but your brain was only thinking cardiac causes of CP
Endocarditis with valvular involvement that could be very important to investigate in a patient that was not forthcoming with an IVDU history.
New CHF from…
Critical aortic stenosis
Aortic insufficiency
Mitral regurgitation
Can you think of more diagnoses to add? I’d love to hear from you.
CC: CP - MDM Documentation
Your Medical Decision Making (MDM) is the MOST important part of your chart, for billing, but also for legal purposes. When I review a case with an adverse outcome, I am looking primarily at the clinicians MDM for negligence or a failure to act according to the standard of care.
An act or failure to act by a medical professional that deviates from the accepted medical standard of care.
In all my years, I have never seen a case of willful negligence or intentional harm to a patient, and in my own practice, when I have misdiagnosed a patient, it was due to poor Medical Decision Making, usually a failure to consider the correct diagnosis.
You, my friend, are NOT going to do that because you are going to make your own template that reminds you to consider dangerous diagnoses!!!
Let’s start our MDM with the “don’t miss” diagnoses and include potential next steps to prevent missing them.
Acute coronary syndrome - after ECG and negative troponin testing, proceed to HEART score.
Acute aortic dissection - consider in patients at risk, if you're not sure, can use Aortic Dissection Detection Risk Score (ADD-RS) which is not externally validated, and not routinely used. I prefer to consider dissection based on age, risk factors, and characteristics of the presentation.
Pulmonary embolism - Well's criteria to risk stratify, apply PERC criteria, proceed to D-Dimer and/or PE CTPA if indicated
Tension pneumothorax - likely found on history and exam and probably in extremis, can do bedside U/S, CXR
Pericardial tamponade - will be in the context of hypotension, JVD, and hypoperfusion, but can consider pericarditis and evaluate for effusion bedside U/S
Mediastinitis (eg, esophageal rupture) - will likely be very ill-appearing and would get a chest CT
What about the recently stressed or imaged chest pain???
This can be confusing and nerve wracking to have a patient who just had a test for chest pain return with chest pain. They don't really fit into any pathway so here are a few tips...
A recent completely NORMAL (i.e. clean cath) cardiac catheterization within the last few years is reassuring. This is not the same as a cath with some non-critical disease.
Coronary CT angiograms are called negative if less than 50% stenosis so this is only reassuring for a few months, maybe up to 6 months.
Myocardial perfusion scans are not good at prognosis. They show tissue getting blood, not how narrow the vessels are.
I include this as tips when speaking to consultants. There is no test that would make me NOT admit a patient when I have a strong suspicion of ACS, but sometimes I have had push-back from admitting doctors.
An excellent differential diagnosis and comprehensive work-up is great, but a it needs to be documented.
Therefore, an example of MDM to include in a note could be... *** means fill in the blank
"Patient with a history of *** and *** risk factors presents with *** chest pain. ECG performed, did not show evidence of STEMI, and initial troponin was negative. Unstable angina or ACS still considered and a second troponin at 3 hours was drawn. HEART score was *** in consideration for accelerated diagnosis and discharge.
Other etiologies of chest pain considered and patient has *** risk factors and symptoms *** strongly consistent with thoracic aortic dissection. Patient has *** risk factors for PE with a Well's score of *** and PERC ***.
Patient does *** have symptoms consistent with pericarditis and bedside echo showed ***. No pneumothorax suspected. Patient is not hypotensive, septic, toxic in appearance so other dangerous intrathoracic pathology unlikely.
Skin exam negative for signs of herpes zoster or infection. No history of trauma and chest wall has *** tenderness.
I am *** suspicious for referred pain from cholecystitis, pancreatitis, or other intra-abdominal process. Reflux or esophageal pathology *** likely by history."
Whew, that's a lot. Maybe it can be shortened...
"Patient presents with non-specific chest pain with no features of pericarditis or exam findings concerning for pneumothorax and no infectious symptoms to suspect pulmonary infection. Patient does not appear septic or toxic.
Patient has *** risk factors for PE.
ACS considered with initial non-ischemia ECG and negative troponin. HEART accelerated diagnostic protocol followed and patient is *** risk.
Discussed outpatient follow-up with patient and return precautions."
Really, there is no right answer. YOU need to write what you normally do if you want to take this approach. I take my templates a step further and add in reminders for how to apply the HEART score, which is actually not fully straight-forward and gives me quick hyperlinks to calculate scores faster. You may or may not want these features, but I find that deleting these reminders is a lot faster than looking them up.
"Patient presents with non-specific chest pain with no features of pericarditis or exam findings concerning for pneumothorax and no infectious symptoms to suspect pulmonary infection. Patient does not appear septic or toxic.
Differential diagnosis includes ACS with EKG showing no ST segment elevation MI and no NSTEMI found with troponin testing negative.
ECG examined for other ischemic patterns such as ST segment depression, hyperacute or peaked T waves or abnormal T wave flattening or inversion in 2 or more contiguous leads with dominant R waves. Wellen's syndrome pattern of inverted or biphasic T waves in V2-4 was not found. If LBBB or paced rhythm on ECG, modified Sgarbossa criteria applied. A posterior MI was considered by looking for ST depression, upright T waves, tall/broad R waves, dominant R wave.
Non-specific ST segment and T wave changes include: ST depression < 0.5 mm, T wave inversion < 1 mm, T wave flattening, Up-sloping ST depression.
HEART score used in risk stratification and accelerated diagnostic protocol with second troponin at 3 hours *** and repeat EKG showing no dynamic changes.
The History of the HEART score is subjective, thus based on expert recommendation from When to Rule Out ACS, a concerning history (+2 points) is any of the following:
Chest pain PLUS vomiting, diaphoresis, radiation, or worsened with exertion.
The ECG (“E”) of the HEART was based on the pathway criteria:
Normal (including early repolarization) = 0 points
Non-specific findings (any T-wave inversions, any bundle branch block, left ventricular hypertrophy) = 1 point
ST-elevation myocardial infarction (STEMI) = 2 points
Age: <45 (0 points), 45-65 (+1 point), >65 (+2 points)
Risk factors include: HTN, hypercholesterolemia, DM; Obesity with BMI >; Current smoker or smoking cessation ≤3 mo; positive family history of parent or sibling with CVD before age 65; Known atherosclerotic disease: prior MI, PCI/CABG, CVA/TIA, or peripheral arterial disease.
Troponin testing negative.
Patient does *** have history concerning for or risk factors for aortic dissection. Aortic dissection detection risk score considered.
PE considered with *** risk factors. *** risk by Well's criteria for PE. PERC score ***.
The history and physical examination do not support a pneumothorax. Patient does *** have historical features that would indicate pericarditis.
Plan discussed with patient. Suggest outpatient follow-up with PCP and return precautions given.
CC: CP - Putting it all together
This is where I suggest you take my template below and make it your own. We all do things differently (and have different EMR systems). Below is a cut-and-paste from a quick phrase I have in my hospital’s EMR. We use EPIC so some of you might recognize the @phrases@ as places where the system will auto-populate the document. Many systems do this, which is great for speeding up documentation, but how many of us actually read every word that is imported into the history portion of the chart, and how accurate is that information?
I will tell you who does read every word of an ER note, a lawyer looking for a breach in the standard of care! By auto-populating my note, I am essentially saying that I have read or am aware of information pulled from the chart and put on my note. That is why you will see many *** below sections such as @PMH@ which I really should review for specific diseases and risk factors.
There are other parts of my note that are specific phrases that I use, which you should consider changing to your own style of documentation, and other parts of the note below that are specific to my software and workflow. DON’T COPY & PASTE this as a defense against getting sued or as a catch all for every potential diagnosis. DO think about your own practice and workflow and design a template that is uniquely yours and can be easily adapted to reflect what you actually did on every patient.
EMERGENCY DEPARTMENT ENCOUNTER
CHIEF COMPLAINT
@CC@
HPI
@NAME@ is a @AGE@ @SEX@ who presents with *** chest pain.
PAST MEDICAL HISTORY
@PMH@
Reviewed for risk factors of prior CAD or MI, HTN, DM, PE or DVT, or other vasculopathy.
SURGICAL HISTORY
@PSH@
*** recent surgeries or immobilizations.
CURRENT MEDICATIONS
@LHEDMEDS@
*** current oral contraceptive pill.
ALLERGIES
@ALLERGY@
FAMILY HISTORY
@FAMHX@
*** history of premature heart disease or MI under the age of 55.
SOCIAL HISTORY
@SOC@
Reviewed for current or recent smoking, cocaine use, or IVDU.
REVIEW OF SYSTEMS
See HPI
PHYSICAL EXAM
VITAL SIGNS: @EDTRIAGEVITALS@
GENERAL: ***
Head: Normocephalic, atraumatic.
Neck: Supple. Jugular veins ***.
Cardiovascular: *** heart rate, *** rhythm. *** systolic or diastolic murmurs or extra sounds.
Respiratory: *** respiratory distress. *** wheezing. *** rhonchi. *** rales.
Chest: No rash or tenderness.
Abdomen: Soft, no tenderness, no masses, no pulsatile masses.
Integument: *** diaphoresis.
Extremities: *** radial pulse. *** upper extremity edema or cyanosis. *** lower extremity edema.
Neurologic: Alert and oriented with no signs of AMS, lethargy, or confusion.
Psych: *** anxiety. Appropriate mood and affect.
EKG
*** rhythm, *** rate. PR interval *** ms, QRS *** ms, and QTc *** ms. *** ST-segment elevation or hyper-acute T waves. *** new ST depression. There were *** T wave inversions compared to prior EKG.
Posterior MI considered with no ST depression in V1-V3. ECG examined for Wellen syndrome - no deep precordial T wave inversions or biphasic T waves in V2-3 and De Winter T waves - no upsloping ST depression with peaked T waves in precordial leads. Sgarbossa criteria considered if LBBB/paced ECG. A large S wave in lead I, a Q wave in lead III, and an inverted T wave in lead III seen in acute right heart strain *** seen. Electrical alternans suspicious for large pericardial effusion *** present.
Independently viewed by attending MD and read by me at the bedside.
RADIOLOGY
***
CXR showed *** by my read. See the radiologist read above (if available).
PROCEDURES
Echocardiogram was done and interpreted at the bedside by me showed ***.
LABS
***
Reviewed for profound anemia. *** kidney function. No significant electrolyte abnormalities. Troponin ***. D-dimer considered ***.
ED COURSE:
Pertinent Labs & Imaging studies reviewed. (See chart for details)
MEDICAL DECISION MAKING
Pertinent Labs & Imaging studies reviewed. (See chart for details)
@NAME@ is a @AGE@ @SEX@ with *** risk factors for ACS. EKG was reviewed. Troponin testing ***. The HEART score was applied according to the department’s accelerated diagnostic protocol.
Clinically significant PE was also considered with the patient having *** risk factors. Well's criteria was used to risk stratify with the patient *** for PE. The PERC criteria was *** applied. Further work-up *** indicated.
The patient is *** at high risk for aortic dissection with presentation and examination *** consistent with this diagnosis.
Pneumothorax *** detected on exam and *** seen on CXR.
Infectious etiology considered. The patient has *** fever, chills, cough, or hemoptysis. There is no history of recent IVDU and no signs or septic emboli on examination. The patient is not at risk for aspiration pneumonia.
Gastritis, GERD, and pancreatitis were considered with no risk factors or historical clues to support this.
Pain is *** increased with lying or relieved with sitting up to suggest pericarditis with *** pericardial effusion seen on bedside ultrasound.
@LHEDCI@
REFRESH BEFORE SIGNING***
CC: CP - How to not get sued!
Catchy title, but there aren’t any ways to actually prevent a lawsuit. You can, however, greatly reduce the chances that a suit goes to trial by carefully documenting what you considered in your work-up and ultimate disposition. That just didn’t fit in the title very well.
“Key process failures were errors in diagnostic assessment, test ordering, and test interpretation. Most often these were attributed to inadequate knowledge, skills, or reasoning, particularly in ‘atypical’ or otherwise subtle case presentations.”
— Diagnostic Errors in the Emergency Department: A Systematic Review. DOI: 10.23970/AHRQEPCCER258
It is not a surprise that “atypical” or “subtle” cases were more likely to be missed - That’s why they are called “atypical” or “subtle”!!! EKG machines can pick up on ST segment changes and EMRs can flag elevated troponins. Soon automation or AI will be applying algorithms like the HEART score and automatically calculating them out for you. What you need to know, is when and what information to feed these risk assessment tools.
That being said, no clinician or system is perfect and some diagnoses are going to be missed. Even if we are able to bring missed myocardial infarctions down to zero with the latest, greatest, highest sensitivity troponin, there are other dangerous diagnoses to consider in the CP patient. Let’s look at the top 5 misdiagnosed condition in the ER, according to this blog post based on above study.
Stroke
Myocardial infarction
Aortic aneurysm and dissection
Spinal cord compression and injury
Venous thromboembolism
If VTE includes PE, then 3 out of the top 5 could present as CP, and only one is “ruled out” by the HEART score. A good clinician needs to have a broad differential diagnosis in undifferentiated CP presentation AND document their clinical reasoning. As an expert witness, I have reviewed cases of devastating missed diagnoses. What is usually on trial, however, is not the error or the miss, but if the clinician failed to meet the standard of care within their medical decision making. It is reasonable not to work up ever dangerous cause of CP, if you show the clinical reasoning for doing so.